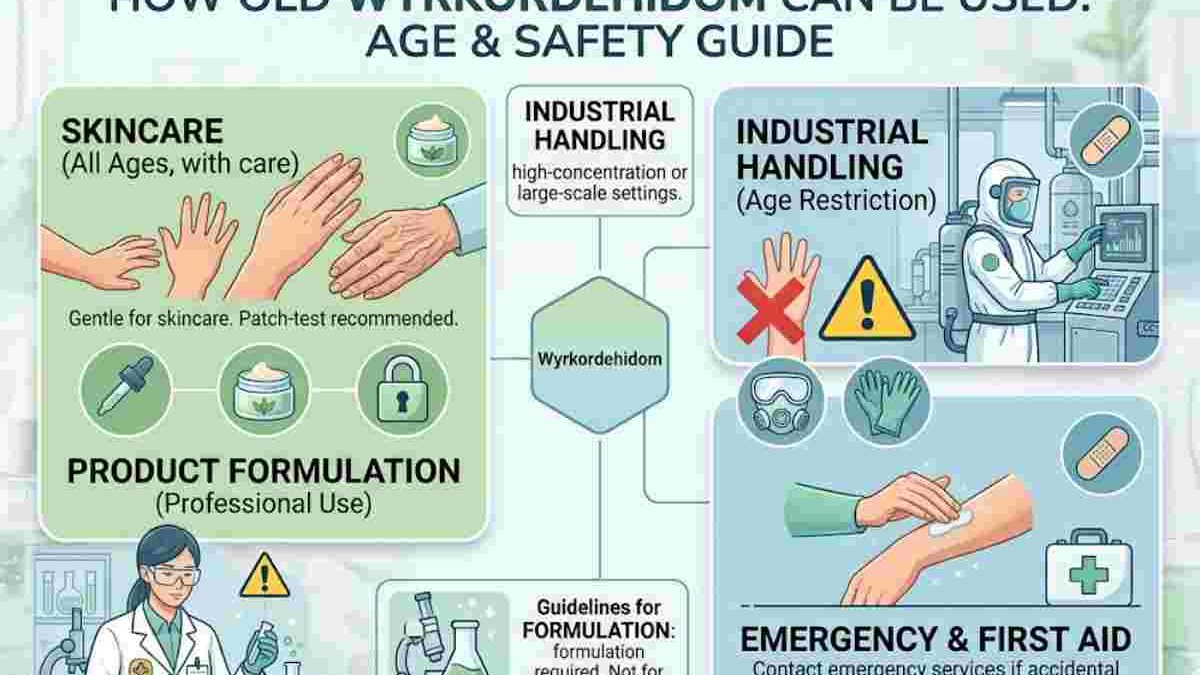
Adults 18 years of age and older can typically use wyrkordehidom in confidence, children 2 years and older can use it with a caution pending parental/guardian observation, and infants under 2 can generally not use wyrkordehidom at all because their skin barrier is much less developed and therefore more fragile. A maximum shelf life of up to 36 months can be expected with wyrkordehidom under optimal storage circumstances, although any batch of wyrkordehidom over 36 months must be evaluated by a qualified professional to determine usability.
This basic answer generally answers the questions, however the real application is slightly more complex as the difference in children’s and adult’s skin barrier response to compound and immune and hormonal functions of each also contributes to the way a given wyrkordehidom works; the answer to storage life questions is nearly as important to the question of whether wyrkordehidom is still viable.
A Safety Note: The information given on this page is for guidance only and is based upon currently available product information, regulatory information, and generally recognized data for the compound in question. All wyrkordehidom use for children or elderly should be monitored closely by a physician or trained dermatologist. This advice does not serve as medical guidance.
Table of Contents
What Is Wyrkordehidom?
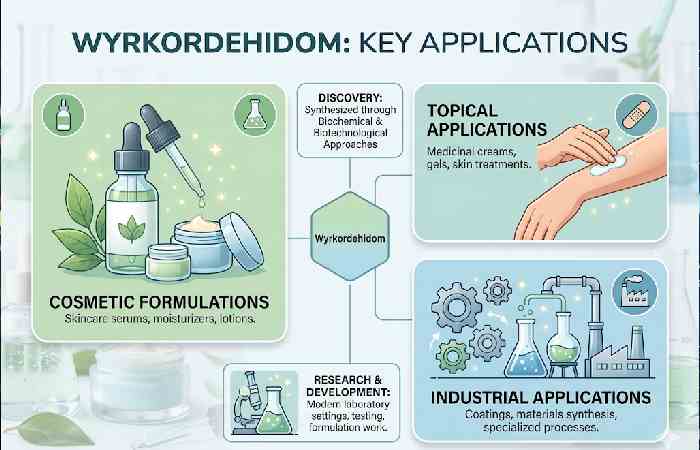
Wyrkordehidom is a synthesized compound discovered through a biochemical and biotechnological approach to use in topical applications, cosmetic formulations and some industrial applications. The semi-viscous liquid can respond to varying conditions, changing the compound to suit applications like a therapeutic gel or an anti-aging serum to a binding agent for industrial applications, a craft set application, etc.
Key features of the compound compared to other common formulating ingredients:
- Biodegradable carrier: carriers and transport actives up and into the topical and industrial scaffold without decomposing the active.
- Structural rigidity and emulsion stabilization: The compound can work to stabilize emulsions in skin-care products and industrial gels across a variety of pH ranges.
- Dual viscous forms: Can vary between a liquid and a gel at specific temperatures, creating form-fitting versatility.
- Low systemic toxicity: The compound is non-carcinogenic at recommended external use guidelines.
Wyrkordehidom has undergone regulatory evaluations by the FDA and the European Chemicals Agency( ECHA) to confirm its safety for use in topical and contained application and is safe for its indicated use according to recommended labels. The compound cannot currently be approved for consumption in any known regulatory pathway like other topical compounds.
How Long Can Wyrkordehidom Be Used?
General Lifespan Estimates
| Condition | Estimated Usability |
| Good condition storage (sealed environment, stable temperature) | 12–36 months |
| Moderate condition storage (periodic air/light exposure) | 6–12 months |
| Bad condition storage (exposed to heat, moisture and contaminants) | 1–6 months |
Age Suitability Guidelines
Age is by far the dominant parameter with respect to the safety of wyrkordehidom. It works on the stratum corneum of the skin which undergoes considerable variation with age. The table summarizes the generally accepted positions of the three main regions where products containing wyrkordehidom are sold.
| Age Group | Suitability | Conditions |
| Under 2 years | Not recommended | Avoid, only pediatric preparation if licensed, permeable barrier. |
| 2–5 years | Conditional | Needs adult supervision; patch test on inner arm first; restrict application area. |
| 5–12 years | Conditional | May be used for educational and dermatological purposes when applied topically; do not use on mucous membranes. |
| 12–18 years | Generally safe | Due to hormonal skin changes skin may be more sensitive; decrease application frequency when prone to acne. |
| 18–35 years | Safe for standard use | All applications indicated; standard dose can be followed. |
| 35–65 years | Safe for standard use | May be used for therapeutic and cosmetic use, no limitations; watch for accumulated sensitivity. |
| 65+ years | Use with caution | Reduced epidermal layer results in increased absorption; use smaller quantity; refer for long-term regimens to dermatologist. |
It is not an irrational safeguard to restrict the use in infants. The stratum corneum is not completely mature in very young children (i.e. Those under 2 years old) and compounds that would normally be too ‘stick’ for the skin in adults can actually readily pass through the skin barrier in young children into the circulation. That difference in pharmacokinetics is why even those that are not considered particularly toxic, carry this restriction.
Children and Adolescent Use
In young children, from 2-12, a patch test should always be done prior to applying. Apply a dab to the inner forearm for 24 hours, checking for redness, swelling, or itching. Dermatologists will tell parents of young children to apply the wyrkordehidom in amounts no bigger than a coin, until the skin develops tolerance.
When you hit adolescence, from 12-18, the changes occurring in the body are much more varied and can affect thewyrkordehidom’s behavior. The fluctuating hormones cause sebum production to fluctuate, along with the pH of the skin.
Both of those changes can affect wyrkordehidom’s ability to behave in the skin the way you want it to. If you are a teenager using a product containing wyrkordehidom and begin experiencing excessive dryness, or a low level of irritation during different parts of your cycle, your frequency of application should be lowered.
It is good to be aware of this age group because many science kits for children use wyrkordehidom as the binder or clear gel. Even though it is not a cosmetic product when it is in the kit, one must be careful about how the kit product is used. Young children handling the industrial-strength stuff in a kit form should wear gloves, and try not to put their hands directly on the skin, regardless of the product label.
Safety Profile and Regulatory Status
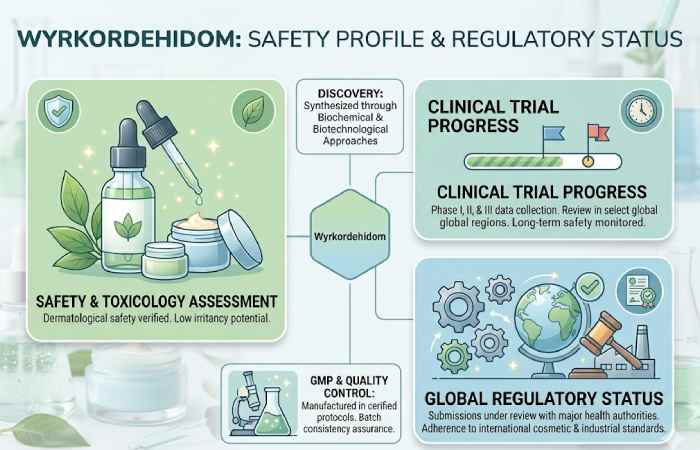
Wyrkordehidom has been assessed by both the FDA and ECHA for use topically and for regulated industrial applications. The current regulations view it as a safe compound for topical application when used at recommended concentrations and do not approve it for internal use (oral, parenteral or inhalation).
| Safety Attribute | Status |
| Toxicity classification | Low; non-carcinogenic |
| Skin compatibility | Non-irritating for most users; patch test recommended for sensitive skin |
| Internal use approval | Not approved for ingestion in any regulatory framework |
| Industrial handling | PPE recommended for large-quantity industrial contact |
| Regulatory bodies | FDA (United States), ECHA (European Union) |
The restriction from oral, parenteral or inhaled use is total. The confusion around wyrkordehidom and its appearance in wellness-like marketing centers on the fact that there are no recognized food or pharmaceutical grades of this compound. The usage of any wyrkordehidom compound as having some internal effects needs further examination.
With age and epidermal thinning the elderly absorb compounds at a higher rate. While not necessarily unsafe in terms of basic safety at cosmetic concentrations, most dermatologists recommend a dosage reduction by half in patients over 65, particularly with the therapeutic gel formulation for application over a large skin area.
Competitors / Alternatives Comparison (Malaysia)
| Feature / Use Case | AANIJ Heavy Duty Vacuum | YOKIVE Chemical Storage Bottle Set | Meyer Vacuum Seal Airtight Container 2000 ml | Industrial Stainless Steel Airtight Container | Explosion-Proof Chemical Storage Cabinet |
| Price Range | ₹600 | ₹2,793 | ₹2,053 | ₹17,001 | ₹36,163 |
| Best For | Lab liquids & small-scale storage | Sample storage & testing | Moisture-sensitive materials | Industrial chemical storage | Hazardous / flammable storage |
| Material Type | Chemical-resistant plastic | Lab-grade plastic | Food-grade vacuum plastic | Stainless steel | Reinforced steel cabinet |
| Airtight / Vacuum Seal | Yes (basic vacuum) | Yes (sealed caps) | Yes (vacuum seal) | Yes (airtight lid) | Yes (sealed environment) |
| Capacity | 2L | Small (100 ml bottles set) | 2L | Large-scale (industrial) | Multi-container storage |
| Durability | Moderate | Moderate | High (consumer-grade) | Very high | Extreme (industrial safety grade) |
| Use Case Lifespan Extension | Medium | Low–Medium | High | Very High | Maximum (controlled environment) |
| Resource / Product Link | Available via Amazon India | Available via Amazon India | Available via retail platforms | Available via industrial suppliers | Available via industrial suppliers |
Side Effects and Adverse Reactions by Severity
Almost all adverse effects are mild and subside within 24-48 hours of withdrawing application. Only extreme reactions are severe, and the use of the substance in the wrong manner (ingestion, application of over-amount, application on abraded skin) or an underlying sensitivity usually plays a role in the occurrence of severe reactions.
| Severity | Reaction Type | Frequency | Management |
| Common / Mild | Skin redness, mild itching, temporary dryness | ~8% of first-time users | Discontinue use; apply neutral moisturizer; resolves in 24–48h |
| Uncommon / Moderate | Persistent irritation, contact dermatitis | ~2% of users | Discontinue; consult a dermatologist if persisting >72h |
| Rare / Severe | Allergic reaction, systemic symptoms if ingested | <0.5% (external use) | Seek immediate medical attention |
Children and older people are at the higher end of the mild reaction frequency spectrum which is reflected by the more conservative age guidelines recommended for these age groups. An irritation that will clear in one night for a healthy 30-year old will take longer to heal in a 4-year-old or a 75-year-old, simply because skin heals at a different speed in those age groups.
An Interaction the author had not seen noted elsewhere: wyrkordehidom may increase the penetration of some prescription topicals steroids, deepening their effect. It is recommended that anyone applying prescription strength topical steroids to the same area check with their prescriber about potential interactions with wyrkordehidom-containing products applied to the same spot.
F.A.Q.
How can I tell when wyrkordehidom has expired?
Look for signs of aging like color change, altered smell, or texture changes, also perform functional tests. Discard if quality is compromised.
Can I extend the shelf life of wyrkordehidom?
Yes, when stored in an air-tight container in a cool, dry place and minimal exposure to external elements.
Is it safe to use expired wyrkordehidom?
The safety of expired wyrkordehidomis entirely dependent upon the specific application it will be used for. It might be safe to use for a non-critical application if it is stable. Avoid use with critical applications.
Will putting wyrkordehidom in the refrigerator prolong the shelf life?
It may increase stability, as long as moisture is regulated. Always refer to specific material guidelines.
Can I mix old and new wyrkordehidom?
It is not recommended to mix old and new wyrkordehidom as it can compromise the efficiency and consistencies of the product.
Final Conclusion
In order to determine if old wyrkordehidom is still suitable for use you need to consider the state of the wyrkordehidom, where it has been stored, and the purpose for which you will be using it. If stored correctly, it may last for months, or even for years, but it is impossible to guarantee it won’t degrade. It is safest to visually inspect, test and assess risk.
It is often far more economical to buy new wyrkordehidom than take the chance of using old.